3704. α-Estradiol
Nomenclature
CAS number: 57-91-0
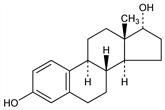
(17α)-Estra-1,3,5(10)-triene-3,17-diol; 1,3,5-estratriene-3,17α-diol; 3,17-dihydroxyestratriene. C18H24O2; mol wt 272.38.
C 79.37%, H 8.88%, O 11.75%.
Description and references
Has been isolated from pregnancy urine of mares. Prepn from β-estradiol by inversion of the hydroxyl group at C-17 after tosylation: Allais, Hoffmann, US 2835681 (1958).
Properties
Needles with 1/2 H2O from 80% alcohol, mp 220-223°. [α]D20 +53 to +56° (c = 0.9 in dioxane). Not precipitated by digitonin (in 80% alcoholic soln). Soluble in alcohol, acetone, aq alkalies. One gram dissolves in more than 100 ml of boiling benzene. Slightly sol in ether, chloroform. Insol in water, aq dil acids.Derivative
Diacetate.C22H28O4; mol wt 356.46.
C 74.13%, H 7.92%, O 17.95%.
Properties
mp 140-142°.Derivative
3-Benzoate.C25H28O3; mol wt 376.49.
C 79.75%, H 7.50%, O 12.75%.