3707. Estriol
Nomenclature
Description and references
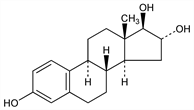
A metabolite of, and considerably less potent than the hormone estradiol, q.v. It is usually the predominant estrogenic metabolite found in urine. During pregnancy the placenta produces relatively large amounts of estriol. Isoln from human pregnancy urine: Marrian, Biochem. J. 23, 1090, 1233 (1929); probably occurs as a glycuronide: Cohen, Marrian, ibid. 29, 1577 (1935). Isoln from human placenta: Collip, Br. Med. J. II, 1080 (1930); Collip et al., Endocrinology 18, 71 (1934). Also obtained from plant sources. Isoln from pussywillows: Skarzynski, Nature 131, 766 (1933). Structure: Huffman, Lott, J. Am. Chem. Soc. 69, 1835 (1947). Crystal and molecular structure: Cooper et al., Acta Crystallogr. 25B, 814 (1969). Soly studies: Ruchelman, Howe, J. Chromatogr. Sci. 7, 340 (1969). Partial synthesis: Huffman, J. Biol. Chem. 169, 167 (1947). Syntheses: Huffman, Lott, J. Am. Chem. Soc. 71, 719 (1949); Leeds et al., ibid. 76, 2943 (1954).
Properties
Very small, monoclinic crystals from dil alc. d 1.27. mp 282°. During heating on the microscope heating stage, rearrangement of the crystal structure takes place at 270° and 275° (rate of heating, 4°/min, Kofler microscope heating stage). [α]D25 +58° ±5° (0.04 g in 1 ml dioxane). uv max: 280 nm. Precipitated by digitonin. Practically insol in water. Sol in alcohol, dioxane, chloroform, ether, vegetable oils; freely sol in pyridine, in solns of fixed alkali hydroxides.Derivative
Triacetate.Properties
Crystals from 90% alc. mp 126°.Derivative
16,17-Bis(sodium hemisuccinate).Nomenclature
Description and references
Prepn: GB 879014 (1960 to Organon).
Therapeutic Category
Estrogen.
Therapeutic Category (Veterinary)
Estrogenic hormone therapy.Keywords
Estrogen; Steroidal