3720. Ethambutol
Nomenclature
CAS number: 74-55-5
2,2′-(1,2-Ethanediyldiimino)bis-1-butanol; (+)-2,2′-(ethylenediimino)di-1-butanol; d-N,N′-bis(1-hydroxymethylpropyl)ethylenediamine; EMB. C10H24N2O2; mol wt 204.31.
C 58.79%, H 11.84%, N 13.71%, O 15.66%.
Description and references
Prepn: Wilkinson et al., J. Am. Chem. Soc. 83, 2212 (1961); eidem, J. Med. Chem. 5, 835 (1962). Pharmacology: Kulig et al., Diss. Pharm. Pharmacol. 23, 463 (1971), C.A. 76, 81239d (1972). Comprehensive description: C. S. Lee, L. Z. Benet, Anal. Profiles Drug Subs. 7, 231-249 (1978). Mechanism of action: W. H. Beggs in Antibiotics vol. 5(pt. 1), F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 43-66.
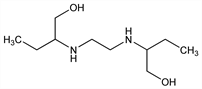
Properties
mp 87.5-88.8°. [α]D25 +13.7° (c = 2 in water). Sol in chloroform, methylene chloride; less sol in benzene; sparingly sol in water.Derivative
Dihydrochloride.Nomenclature
CAS number: 1070-11-7
Dexambutol (L'Arguenon); Ebutol (Kaken); Etibi (Gerot); Etapiam (Piam); Myambutol (Wyeth); Mycobutol (Cadila); Sural (Chinoin); Tibutol (Bracco). C10H24N2O2.2HCl; mol wt 277.23.
C 43.32%, H 9.45%, N 10.10%, O 11.54%, Cl 25.58%.
Properties
mp 198.5-200.3°; also reported as mp 201.8-202.6°. [α]D25 +7.6° (c = 2 in water). Sol in water, DMSO; sparingly sol in ethanol; difficultly sol in acetone, chloroform.Derivative
Mixture with isoniazid methane sulfonate.Nomenclature
CAS number: 41663-50-7
(+)-2,2-(Ethylenediimino)dibutanol diisoniazide methanesulfonate; isobutol; Isoetam (Ferrer). Description and references
Prepn: C. Ferrer-Salat et al., BE 753862; US 3718655 (1971, 1973 both to Lab. Ferrer).
Properties
White powder, crystallizes as needles, mp 121-122° (dec). Sol in water, methanol, N,N-dimethylformamide. Insol in ethanol, ethyl ether, chloroform. uv max: 265 nm. [α]D20 +3° (c = 5% in water). LD50 in mice (g/kg): 2.800 orally; 2.210 i.p. (Ferrer-Salat).Therapeutic Category
Antibacterial (tuberculostatic).
Keywords
Antibacterial (Tuberculostatic)