3860. Ethynodiol
Nomenclature
CAS number: 1231-93-2
(3β,17α)-19-Norpregn-4-en-20-yne-3,17-diol; 17α-ethynyl-19-norandrost-4-ene-3β,17β-diol; 17α-ethynyl-4-estrene-3β,17β-diol; ED. C20H28O2; mol wt 300.44.
C 79.95%, H 9.39%, O 10.65%.
Description and references
Prepn: F. B. Colton, US 2843609 (1958 to Searle). Prepn of the 3-acetate, 17-acetate, and diacetate: P. D. Klimstra, US 3176013 (1965 to Searle); see also F. Sondheimer, Y. Klibansky, Tetrahedron 5, 15 (1959). Pharmacokinetics and metabolism: C. J. Lewis et al., Xenobiotica 10, 705 (1980). Comparative clinical study in oral contraceptives: M. H. Briggs, J. Reprod. Med. 28, Suppl. 1, 92 (1983). Review of carcinogenicity studies: IARC Monographs 21, 387-398 (1979). Comprehensive description: E. P. K. Lau, J. L. Sutter, Anal. Profiles Drug Subs. 3, 253-279 (1974).
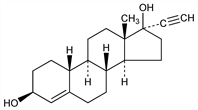
Derivative
Diacetate.Nomenclature
CAS number: 297-76-7
3β,17β-Diacetoxy-17α-ethynyl-4-estrene; SC-11800; Femulen (Searle); Luteonorm (Serono); Luto-Metrodiol; Metrodiol (Clin-Comar-Byla). C24H32O4; mol wt 384.51.
C 74.97%, H 8.39%, O 16.64%.
Properties
Crystals from methanol + water, mp ≈126-127°. [α]D -72.5° (chloroform).Derivative
Diacetate mixture with mestranol.Nomenclature
Luteolas (Serono); Metrulen; Ovaras (Serono); Ovulen.Derivative
Diacetate mixture with ethinyl estradiol.Nomenclature
Conova (Gold Cross); Demulen (Searle); Miniluteolas (Serono).Therapeutic Category
Progestogen; in combination with estrogen as oral contraceptive.
Keywords
Contraceptive (Oral); Progestogen