3863. Etidronic Acid
Nomenclature
Description and references
Bisphosphonate antiresorptive agent. Prepn: H. von Baeyer, K. A. Hofmann, Ber. 30, 1973 (1897); and characterization of the acid and disodium salt: F. Kasparek, Monatsh. Chem. 99, 2016 (1968); B. Blaser et al., Z. Anorg. Allg. Chem. 381, 247 (1971). Use as detergent builder: F. L. Diehl, US 3159581 (1964 to Proctor & Gamble). Determn in pharmaceutical formulations: E. W. Tsai et al., J. Pharm. Biomed. Anal. 11, 513 (1993). Symposium on clinical efficacy in malignancy-related hypercalcemia: Am. J. Med. 82, Suppl 2A, 1-78 (1987). Review of pharmacology and therapeutic efficacy in resorptive bone disease: C. J. Dunn et al., Drugs Aging 5, 446-474 (1994); of clinical safety and efficacy in osteoporosis: T. P. van Staa et al., Pharmacotherapy 18, 1121-1128 (1998).
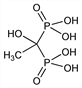
Properties
Crystallizes from water as the monohydrate. pK1 1.35 ±0.08; pK2 2.87 ±0.01; pK3 7.03 ±0.01; pK4 11.3. Very sol in water (69% at 20° C). Insol in acetic acid.Derivative
Disodium salt.Nomenclature
Properties
Crystallizes from water as the di- or tetrahydrate.Use
Sequestering and chelating agent; scale and corrosion inhibitor.Therapeutic Category
Bone resorption inhibitor.
Keywords
Antiosteoporotic; Bone Resorption Inhibitor