3888. Etorphine
Nomenclature
CAS number: 14521-96-1
[5α,7α(R)]-4,5-Epoxy-3-hydroxy-6-methoxy-α,17-dimethyl-α-propyl-6,14-ethenomorphinan-7-methanol; tetrahydro-7α-(1-hydroxy-1-methylbutyl)-6,14-endo-ethenooripavine; 7,8-dihydro-7α-[1(R)-hydroxy-1-methylbutyl]-O6-methyl-6,14-endo-ethenomorphine; 7α-[1(R)-hydroxy-1-methylbutyl]-6,14-endo-ethenotetrahydrooripavine; 19-propylorvinol; tetrahydro-7α-(2-hydroxy-2-pentyl)-6,14-endo-ethenooripavine. C25H33NO4; mol wt 411.53.
C 72.96%, H 8.08%, N 3.40%, O 15.55%.
Description and references
Narcotic analgesic. Prepn: BE 618392; K. W. Bentley, GB 937214 (1962, 1963 both to J. F. MacFarlan); and structure: K. W. Bentley, D. G. Hardy, Proc. Chem. Soc. London 1963, 220. Improved prepn: W. R. Hydro, US 3763167 (1973 to U.S. Army). Determn by HPLC: I. Jane, A. McKinnon, J. Chromatogr. 323, 19 (1985). Analgesic activity: G. F. Blane, J. Pharm. Pharmacol. 19, 367 (1967). Pharmacology: Lister, ibid. 16, 364 (1964). Use as tranquilizer in dogs: J. L. Crooks et al., Vet. Rec. 87, 498 (1970); M. Grange et al., Rev. Med. Vet. 124, 899 (1973); in large animals: A. M. Harthoorn, J. Am. Vet. Med. Assoc. 149, 875 (1966); B. T. Alford et al., ibid. 164, 702 (1974).
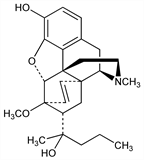
Properties
White crystals from aqueous ethoxyethanol, mp 214-217°. (7β-isomer, melts at 280°).Derivative
Hydrochloride.Nomenclature
M-99.C25H33NO4.HCl; mol wt 447.99.
C 67.03%, H 7.65%, N 3.13%, O 14.29%, Cl 7.91%.
Properties
mp 266-267°. (7β-isomer hydrochloride, dec at 290)°.Derivative
Hydrochloride in combination with acepromazine.Nomenclature
Immobilon (Reckitt & Colman).Derivative
3-Acetate.Nomenclature
Acetorphine; O3-acetyl-7,8-dihydro-7α-[1(R)-hydroxy-1-methylbutyl]-O6-methyl-6,14-endo-ethenomorphine; 3-O-acetyl-7α-[1(R)-hydroxy-1-methylbutyl]-6,14-endo-ethenotetrahydrooripavine; 3-O-acetyl-17-propylorvinol.C27H35NO5; mol wt 453.57.
C 71.50%, H 7.78%, N 3.09%, O 17.64%.
Properties
Crystals from methanol, mp 195°.Derivative
3-Acetate hydrochloride.Nomenclature
M-183.C27H35NO5.HCl; mol wt 490.03.
C 66.18%, H 7.40%, N 2.86%, O 16.32%, Cl 7.23%.