Nomenclature
CAS number: 24159-07-7
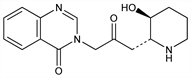
3-[3-[(2
R,3
S)-3-Hydroxy-2-piperidinyl]-2-oxopropyl]-4(3
H)-quinazolinone; 3-[3-(3-hydroxy-2-piperidyl)acetonyl]-4(3
H)-quinazolinone; 3-[β-keto-γ-(3-hydroxy-2-piperidyl)propyl]-4-quinazolone; β-dichroine.
C
16H
19N
3O
3; mol wt 301.34.
C 63.77%, H 6.36%, N 13.94%, O 15.93%.
Description and references
Alkaloid with antimalarial properties; active
component in traditional Chinese medicines. Isoln from Dichroa
febrifuga Lour., Saxifragaceae: J. B. Koepfli et al., J. Am. Chem. Soc. 69, 1837 (1947); eidem, ibid. 71, 1048 (1949); from the common hydrangea:
F. Ablondi et al., J. Org. Chem. 17, 14 (1952). Identity with β-dichroine: Fu, Yang, C.A. 43, 1530a (1949). Structure:
J. B. Koepfli et al., J. Am. Chem.
Soc. 72, 3323 (1950). Synthesis: B. R. Baker et al., J. Org. Chem. 17, 132 (1952); 18, 178 (1953). Config: Hill,
Edwards, Chem. Ind. (London) 1962, 858. Revised stereochemistry: D. F. Barringer, Jr. et al., J. Org. Chem. 38, 1937 (1973); and asymmetric synthesis: S. Kobayashi et al., Tetrahedron Lett. 40, 2175 (1999); Y. Takeuchi et al., Tetrahedron 59, 1639 (2003).
Properties
Dimorphic crystals: needles from ethanol, mp 139-140°; from chloroform, mp 154-156°. [α]D25 +6° (c = 0.5 in chloroform); [α]D25 +28° (c = 0.5 in ethanol). Freely sol in methanol + chloroform, water + ethanol; slightly
sol in water, ethanol, acetone, chloroform. Practically insol in
ether, benzene, petr ether. LD50 orally in white mice: 2.5-3.0 mg/kg (Koepfli, 1949).Derivative
Dihydrochloride.
Nomenclature
CAS number: 32434-42-7
C
16H
19N
3O
3.2HCl; mol wt 374.26.
C 51.35%, H 5.66%, N 11.23%, O 12.82%, Cl 18.95%.
Properties
Crystals from 90% ethanol, mp 220-222° (dec). [α]D25 +12.8° (c = 0.8).