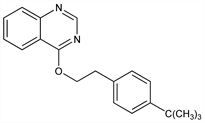
3959. Fenazaquin
Nomenclature
CAS number: 120928-09-8
4-[2-[4-(1,1-Dimethylethyl)phenyl]ethoxy]quinazoline; 4-[2-[4-(tert-butyl)phenyl]ethoxy]quinazoline; 4-tert-butylphenethyl quinazolin-4-yl ether; DE-436; EL-436; Magister (Margarita). C20H22N2O; mol wt 306.40.
C 78.40%, H 7.24%, N 9.14%, O 5.22%.
Description and references
Acaricidal quinazoline; inhibits Complex I of the mitochondrial electron transport chain. Prepn: B. A. Dreikorn et al., EP 326329 (1989 to Lilly); idem et al., US 5411963 (1995 to DowElanco). Mechanism of action study: E. Wood et al., Pestic. Biochem. Physiol. 54, 135 (1996). Photodecomposition study: J. Bhattacharyya et al., J. Agric. Food Chem. 51, 4013 (2003). GC determn in environmental and crop samples: A. R. Gambie, J. M. Perkins, Brighton Crop Prot. Conf. - Pests Dis. 1992, 895. Field trials vs spider mites on ornamentals: R. T. Pollak et al., ibid. 1181. Comprehensive description: C. Longhurst et al., ibid. 51-58.