3978. Fenofibrate
Nomenclature
CAS number: 49562-28-9
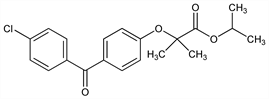
2-[4-(4-Chlorobenzoyl)phenoxy]-2-methylpropanoic acid 1-methylethyl
ester; isopropyl [4′-(p-chlorobenzoyl)-2-phenoxy-2-methyl]propionate; procetofen; procetofene; LF-178; Ankebin (Volpino); Elasterin (Phoenix); Fenobrate (Gramon); Fenotard (Virbac); Lipanthyl (Fournier); Lipantil (Fournier); Lipidil (Ibirn); Lipoclar (Crinos); Lipofene (Teofarma); Liposit (SIT); Lipsin (Fournier); Nolipax (Foscama); Procetoken (Bernabo); Protolipan (Millet); Secalip (Fournier); Tricor (Abbott). C20H21ClO4; mol wt 360.83.
C 66.57%, H 5.87%, Cl 9.83%, O 17.74%.
Description and references
Prepn: A. Mieville, DE 2250327 (1973 to Orchimed), C.A. 79, 53029 (1973), addn to DE 2003430; eidem, US 4058552 (1970, 1977 both to Orchimed). Series of articles on synthesis, metabolism, pharmacology and clinical trials: Arzneim.-Forsch. 26, 885-909 (1976). Toxicity data: R. Sornay et al., ibid. 885. GC-MS of major metabolite in humans: L. F. Elsom et al., J. Chromatogr. 123, 463 (1976). Efficacy in hyperlipidemias: H. B. St"ahelin et al., Praxis 68, 24 (1979). Effect on human biliary lipids: E. M. Grandjean et al., Schweiz. Med. Wochenschr. 109, 601 (1979). Mechanism of action: W. Wülfert et al., Artery 9, 120 (1981).
Properties
Crystals from isopropanol, mp 80-81°. Practically insol in water. Slightly sol in methanol, ethanol. Sol in acetone, ether, benzene, chloroform. LD50 in mice: 1600 mg/kg orally (Sornay).Therapeutic Category
Antilipemic.
Keywords
Antilipemic; Fibrates