3979. Fenoldopam
Nomenclature
CAS number: 67227-56-9
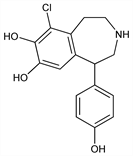
6-Chloro-2,3,4,5-tetrahydro-1-(4-hydroxyphenyl)-1H-3-benzazepine-7,8-diol; 6-chloro-7,8-dihydroxy-1-p-hydroxyphenyl-2,3,4,5-tetrahydro-1H-3-benzazepine; SKF-82526. C16H16ClNO3; mol wt 305.76.
C 62.85%, H 5.27%, Cl 11.60%, N 4.58%, O 15.70%.
Description and references
Dopamine D1-receptor agonist. Prepn: J. Weinstock, DE 2751258; idem, US 4197297 (1978, 1980 both to SmithKline); J. Weinstock et al., J. Med. Chem. 23, 973 (1980). HPLC determn in urine and plasma: V. K. Boppana et al., J. Chromatogr. 317, 463 (1984). Clinical pharmacology: R. M. Stote et al., Clin. Pharmacol. Ther. 34, 309 (1983); R. M. Carey et al., J. Clin. Invest. 74, 2198 (1984). Hemodynamic effects in hypertension: H. O. Ventura et al., Circulation 69, 1142 (1984); M. P. Caruana et al., Br. J. Clin. Pharmacol. 24, 721 (1987). Clinical evaluation in congestive heart failure: G. S. Francis et al., Am. Heart J. 116, 473 (1988).
Derivative
Hydrobromide.C16H16ClNO3.HBr; mol wt 386.67.
C 49.70%, H 4.43%, Cl 9.17%, N 3.62%, O 12.41%, Br 20.66%.
Properties
mp 277° (dec).Derivative
Methanesulfonate.Nomenclature
CAS number: 67227-57-0
Fenoldopam mesylate; SKF-82526-J; Corlopam (SKB). C16H16ClNO3.CH3SO3H; mol wt 401.86.
C 50.81%, H 5.02%, Cl 8.82%, N 3.49%, O 23.89%, S 7.98%.
Properties
mp 274° (dec).Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive; Dopamine Receptor Agonist