3980. Fenoprofen
Nomenclature
Description and references
Prepn: W. S. Marshall, FR 2015728; eidem, US 3600437 (1970, 1971 both to Lilly). Pharmacology: Rubin et al., J. Pharm. Sci. 60, 1797 (1971); 61, 800 (1972); Herrmann, Proc. Soc. Exp. Biol. Med. 139, 548 (1972). Metabolism: Rubin et al., J. Pharmacol. Exp. Ther. 183, 449 (1972). Toxicology: J. L. Emmerson et al., Toxicol. Appl. Pharmacol. 25, 444 (1973). Comprehensive description: C. K. Ward, R. E. Schirmer, Anal. Profiles Drug Subs. 6, 161-182 (1977). Review: R. N. Brogden et al., Drugs 13, 241-265 (1977); R. Nickander et al., in Pharmacological and Biochemical Properties of Drug Substances vol. 1, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1977) pp 183-213.
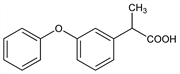
Properties
Viscous oil, bp0.11 168-171°. nD25 1.5742. pKa 7.3.Derivative
Calcium salt dihydrate.Nomenclature
Properties
White crystalline powder. Soly in mg/ml at 37°: n-hexanol 11; methanol 8; water 2.5; chloroform 0.01. pKa 4.5. Aq solns sensitive to intense uv light. LD50 orally in mice: 800 mg/kg (Emmerson).Therapeutic Category
Anti-inflammatory; analgesic.
Keywords
Analgesic; Non-opioids; Acetic/Propionic Acid Derivatives; Anti-inflammatory (Nonsteroidal); Arylpropionic Acid Derivatives