4005. Fenticonazole
Nomenclature
CAS number: 72479-26-6
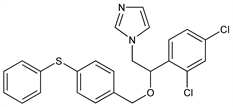
1-[2-(2,4-Dichlorophenyl)-2-[[4-(phenylthio)phenyl]methoxy]ethyl]-1H-imidazole; 1-[2,4-dichloro-β-[[p-(phenylthio)benzyl]oxy]phenethyl]imidazole; 1-(2,4-dichlorophenyl)-2-(N-imidazolyl)ethyl-4-phenylthiobenzyl
ether; 2,4-dichloro-4′-phenylthio-(N-imidazolylmethyl)dibenzyl
ether. C24H20Cl2N2OS; mol wt 455.40.
C 63.30%, H 4.43%, Cl 15.57%, N 6.15%, O 3.51%, S 7.04%.
Description and references
Broad spectrum antimycotic; also active as antibacterial. Prepn: D. Nardi et al., DE 2917244; eidem, US 4221803 (1979, 1980 both to Recordati); eidem, Arzneim.-Forsch. 31, 2123 (1981). Series of articles on physico-chemical properties, in vitro and in vivo antibacterial and antifungal activity, pharmacology and toxicology: ibid. 2127-2154. Toxicity data: G. Graziani et al., ibid. 2145. Preliminary clinical comparison with miconazole in vaginal candidiasis: A. Gastaldi, Curr. Ther. Res. 38, 489 (1985). Comparative clinical trials in dermatomycoses: A. Finzi et al., Mykosen 29, 41 (1986); E. M. Kokoschka et al., ibid. 45.
Derivative
Mononitrate.Nomenclature
CAS number: 73151-29-8
Rec-15-1476; Falvin (Farmades); Fentiderm (Zyma); Fentigyn (Novartis); Lomexin (Recordati). C24H20Cl2NS.HNO3; mol wt 488.41.
C 59.02%, H 4.33%, Cl 14.52%, N 5.74%, S 6.57%, O 9.83%.
Properties
Odorless, white crystalline powder, mp 136°. uv max (methanol): 252 nm (ε 13894). Soly at 20° (mg/ml): water <0.1; ethyl ether <0.1; ethanol 30; methanol 100; chloroform 300; DMF 600. pKa 6.54. LD50 in mice, male rats, female rats (mg/kg): 1191, 440, 309 i.p.; all >3000 orally (Graziani).Therapeutic Category
Antifungal (topical).
Keywords
Antifungal (Synthetic); Imidazoles