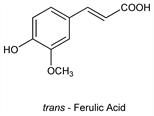
4062. Ferulic Acid
Nomenclature
Description and references
Widely distributed in small amounts in plants. Isoln from Ferula foetida Reg. Umbelliferae: H. Hlasiwetz, L. Barth, Ann. 138, 61 (1866); from Pinus laricio Poir. Abietineae: M. Bamberger, Monatsh. Chem. 12, 441 (1891); see also Beilstein 10, 436 (1927) and supplements. Prepd by the interaction of vanillin, malonic acid and piperidine in pyridine for 3 weeks, then precipitating with HCl: Vorsatz, J. Prakt. Chem. 145, 265 (1936); Pearl, Beyer, J. Org. Chem. 16, 216 (1951). Sepn of cis and trans isomers: Comte et al., Compt. Rend. 245, 1144 (1957). 13C NMR study: C. J. Kelley et al., J. Org. Chem. 41, 449 (1976). Discovery as a component of cell walls in wheat and barley: M. G. Smart, T. P. O'Brien, Aust. J. Plant Physiol. 6, 485 (1979). Use as food preservative: T. Tsuchiya, M. Takasawa, JP Kokai 75 18621 (1975 to Kyokuto Shibosan), C.A. 83, 7602v (1975).