4083. Fingolimod
Nomenclature
Description and references
Immunomodulator evoking a reversible redistribution of lymphocytes from the circulation to secondary lymphatic tissue; synthetic analog of myriocin (ISP-1), a fungal metabolite used in traditional Chinese herbal medicine. Prepn: T. Fujita et al., WO 9408943; eidem, US 5604229 (1994, 1997 both to Yoshitomi); K. Adachi et al., Bioorg. Med. Chem. Lett. 5, 853 (1995); M. Kiuchi et al., J. Med. Chem. 43, 2946 (2000). Efficient synthesis: P. Durand et al., Synthesis 2000, 505. Clinical pharmacokinetics: B. D. Kahan et al., Transplantation 76, 1079 (2003). Clinical evaluation in renal transplant: H. Tedesco-Silva et al., ibid. 77, 1826 (2004). Review of immunopharmacology: P. Troncoso, B. D. Kahan, Clin. Biochem. 31, 369-373 (1998); of mechanism of action: V. Brinkmann, K. R. Lynch, Curr. Opin. Immunol. 14, 569-575 (2002); and pharmacology: T. T. Aki, B. D. Kahan, Expert Opin. Biol. Ther. 3, 665-681 (2003); of development and therapeutic potential: F. J. Dumont, IDrugs 8, 236-253 (2005).
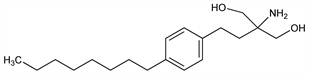
Properties
Crystals from ethyl acetate, mp 103-105°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol, mp 118-120°. Also reported as white solid from ethyl alcohol + ethyl acetate, mp 107-108° (Kiuchi). Sol in water, ethanol. LD50 orally in rats: 300-600 mg/kg (Troncoso).Therapeutic Category
Immunosuppressant.
Keywords
Immunosuppressant