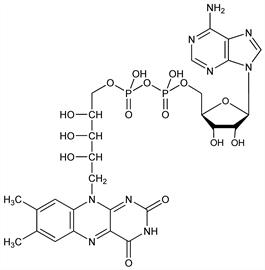
4091. Flavine-Adenine Dinucleotide
Nomenclature
Description and references
The prosthetic group of certain flavoproteins including d-amino acid oxidase, glucose oxidase, glycine oxidase, fumaric hydrogenase, histaminase, and xanthine oxidase. Isoln from yeast: Warburg et al., Biochem. Z. 297, 417 (1938). Structure and isoln from liver, kidneys, hearts, muscles: Warburg, Christian, ibid. 298, 150 (1938); isoln from the mycelium of Eremothecium ashbyii: Yagi, Tada, Biochem. Prep. 7, 51 (1959); Masuda et al., US 2973305 (1961 to Takeda). Synthesis: Christie et al., J. Chem. Soc. 1954, 46; Huennekens, Kilgour, J. Am. Chem. Soc. 77, 6716 (1955); DeLuca, Kaplan, J. Biol. Chem. 223, 569 (1956); Moffatt, Khorana, J. Am. Chem. Soc. 80, 3756 (1958). Review: A. Holmgren, Experientia 36 (Suppl), 149-180 (1980). Review of FAD and other flavin coenzymes: Beinert, The Enzymes vol. 2, P. D. Boyer et al., Eds. (Academic Press, New York, 2nd ed., 1960) pp 339-416; see also vol. XIII Part B (Academic Press, New York, 3rd ed., 1975), several authors.