4111. Floxacillin
Nomenclature
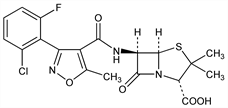
CAS number: 5250-39-5
(2S,5R,6R)-6-[[[3-(2-Chloro-6-fluorophenyl)-5-methyl-4-isoxazolyl]carbonyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 3-(2-chloro-6-fluorophenyl)-5-methyl-4-isoxazolylpenicillin; 6-[3-(2-chloro-6-fluorophenyl)-5-methyl-4-isoxazolecarboxamido]penicillanic
acid; flucloxacillin; BRL-2039. C19H17ClFN3O5S; mol wt 453.87.
C 50.28%, H 3.78%, Cl 7.81%, F 4.19%, N 9.26%, O 17.63%, S 7.06%.
Description and references
Semi-synthetic antibiotic active against penicillin-resistant staphylococci. Halogen-substituted derivative of oxacillin, q.v. Prepn: ZA 6304323 (1964 to Beecham); Nayler, GB 978299; idem, US 3239507 (1964, 1966 both to Beecham). Pharmacology and toxicity: R. Sutherland et al., Br. Med. J. 4, 460 (1970). Clinical studies: Harding et al., Clin. Trials J. 7, 368 (1970); Qureshi et al., ibid. 375.
Derivative
Sodium monohydrate.Nomenclature
CAS number: 34214-51-2
Abboflox (Abbott); Culpen (Fujisawa); Floxapen (GSK); Ladropen (Berk); Stafoxil (Yamanouchi); Staphlipen (Wyeth); Staphylex (GSK). C19H16ClFN3NaO5S.H2O; mol wt 493.87.
C 46.21%, H 3.67%, Cl 7.18%, F 3.85%, N 8.51%, Na 4.66%, O 19.44%,
S 6.49%.
Properties
Freely sol in water. LD50 in mice (g/kg): 2.2 s.c., 3.8 orally (Sutherland).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins