4125. Flucytosine
Nomenclature
Description and references
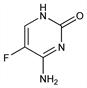
Fluorinated pyrimidine; antifungal activity results from conversion to 5-fluorouracil, q.v., in susceptible fungal cells. Prepn: R. Duschinsky et al., J. Am. Chem. Soc. 79, 4559 (1957); R. Duschinsky, C. Heidelberger, US 2945038 (1960 to Hoffmann-La Roche); Undheim, Gacek, Acta Chem. Scand. 23, 294 (1969). Patents as a fungicide: J. Berger, R. Duschinsky, BE 628615; eidem, US 3368938 (1963, 1968 both to Hoffmann-La Roche). Activity studies: Grunberg et al., Antimicrob. Agents Chemother. 1963, 566; Shadomy et al., ibid. 1968, 452; Grunberg et al., in 5th Int. Congr. Chemother., Proc. vol. IV, K. Spitzy, Ed. (Verlag Wiener Med. Akad., 1967, Austria) p 69. Metabolic studies: B. A. Koechlin et al., Biochem. Pharmacol. 15, 435 (1966). Clinical results: Utz et al., Antimicrob. Agents Chemother. 1968, 344; Warner et al., ibid. 1970, 473. Comprehensive reviews: E. H. Waysek, J. H. Johnson, Anal. Profiles Drug Subs. 5, 115-138 (1976); A. Vermes et al., J. Antimicrob. Chemother. 46, 171-179 (2000).
Properties
Odorless, white crystalline solid, mp 295-297° (dec). uv max (0.1N HCl): 285 nm (ε 8900). Soly in water: 1.5 g/100 ml at 25°C. pKa1: 2.90 pKa2: 10.71. LD50 in mice (mg/kg): >2000 orally and s.c.; 1190 i.p.; 500 i.v. (Grunberg, 1963).Therapeutic Category
Antifungal.
Therapeutic Category (Veterinary)
Antifungal.Keywords
Antifungal (Synthetic)