4153. Fluocortolone
Nomenclature
CAS number: 152-97-6
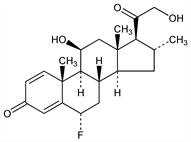
(6α,11β,16α)-6-Fluoro-11,21-dihydroxy-16-methylpregna-1,4-diene-3,20-dione; 6α-fluoro-16α-methyl-1-dehydrocorticosterone; 6α-fluoro-16α-methyl-Δ1,4-pregnadiene-11β,21-diol-3,20-dione; Ultralan oral (Schering AG). C22H29FO4; mol wt 376.46.
C 70.19%, H 7.76%, F 5.05%, O 17.00%.
Description and references
Prepn: BE 614196; Kieslich et al., US 3232839 (1962, 1966 to Schering AG); Doménico et al., Arzneim.-Forsch. 15, 46 (1965); Kieslich et al., Ann. 726, 168 (1969). Pharmacology: Doménico et al., loc. cit; Gillich et al., Int. Z. Klin. Pharmakol. Ther. Toxikol. 1, 197 (1968); Von Schoening et al., Klin. Wochenschr. 48, 1448 (1970). Metabolism: Gerhards et al., Acta Endocrinol. 68, 98 (1971). Clinical studies: Breuer et al., Arzneim.-Forsch. 15, 50 (1965).
Properties
Crystals, mp 188-190.5°. Soly (mg/l): 295 in water (37°); 120 in ethanol (20°); 440 in toluene (20°). [α]D20 +100° (dioxane). uv max (methanol): 242 nm (ε 16300).Derivative
21-Acetate.C24H31FO5; mol wt 418.50.
C 68.88%, H 7.47%, F 4.54%, O 19.12%.
Properties
Crystals, mp 237-239°. uv max (methanol): 240 nm (ε 15860).Derivative
21-Hexanoate.Nomenclature
CAS number: 303-40-2
Fluocortolone 21-caproate; SH-770; Ficoid (Fisons); Ultralanum (Schering AG). C28H39FO5; mol wt 474.60.
C 70.86%, H 8.28%, F 4.00%, O 16.86%.
Properties
Crystals, mp 242-245°. [α]D20 +98.5° (dioxane). uv max (methanol): 242 nm (ε 16200). Soly (mg/l): 7.8 in water (37°); 450 in ethanol (20°); 440 in toluene (20°).Derivative
21-Pivalate.Nomenclature
Fluocortolone trimethylacetate.C22H37FO5; mol wt 400.52.
C 65.97%, H 9.31%, F 4.74%, O 19.97%.
Properties
Crystals, mp 187°. Almost insol in water. Sol in chloroform, methanol. Slightly sol in ether.Therapeutic Category
Glucocorticoid.
Keywords
Glucocorticoid