Nomenclature
CAS number: 144-49-0
2-Fluoroacetic acid; cymonic acid; fluoroethanoic acid; gifblaar poison; monofluoroacetic acid; FAA.
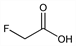
C
2H
3FO
2; mol wt 78.04.
C 30.78%, H 3.87%, F 24.34%, O 41.00%.
Description and references
Toxic constituent of the poisonous South African
plant, Dichapetalum cymosum (Hook.) Engl., Dichapetalaceae, commonly known as gifblaar (“poison leaf”). Also contained in
other poisonous plant species. Metabolically converted to fluorocitrate
which inhibits the tricarboxylic acid cycle resulting in cellular
hypoxia. Prepn via methyl ester: F.
Swarts, Bull. Soc. Chim. [3] 15, 1134 (1896); B. C. Saunders, G. J. Stacey, J. Chem. Soc. 1948, 1773 DOI; by fluorination of disodium malonate: V. Grakauskas, J. Org. Chem. 34, 2446 (1969) DOI. Isoln from gifblaar: J. S. C. Marais, Onderstepoort
J. Vet. Res. 20, 67 (1944). Mechanism
of toxicity: R. A. Peters, Endeavour 13, 147 (1954). Determn in biological samples using HS-SPME: F. Sporkert et al., J. Chromatogr. B 772, 45 (2002) DOI PubMed. Review of properties and use: A. J. Elliott
in Kirk-Othmer Encyclopedia of Chemical Technology vol. 11 (Wiley-Interscience, New York, 4th ed., 1994)
pp 544-550. Review of toxicology: N.
V. Goncharov et al., J. Appl.
Toxicol. 26, 148-161 (2006) DOI PubMed.
Properties
Colorless needles, mp 31-32°. bp 165°. pKa 2.59. d 1.37. Sol in water. Burns with a green flame.Derivative
Sodium salt.
Nomenclature
CAS number: 62-74-8
Sodium fluoroacetate; compound 1080.
C
2H
2FNaO
2; mol wt 100.02.
C 24.02%, H 2.02%, F 18.99%, Na 22.99%, O 31.99%.
Properties
Hygroscopic white solid. mp 200-202° (dec when heated above mp). Soly
at 25° (g/100g): water 111, methanol 5, ethanol 1.4, acetone 0.04,
carbon tetrachloride 0.004.Derivative
Methyl ester.
Nomenclature
CAS number: 453-18-9
Methyl fluoroacetate; MFA.
C
3H
5FO
2; mol wt 92.07.
C 39.14%, H 5.47%, F 20.63%, O 34.75%.
Properties
Mobile liquid, fp 32°. Practically odorless
or faint, fruit-like odor. d420 1.1744; d415 1.1613. nD20 1.3679. bp 104.5°. Sol in water; slightly sol in petr ether. LD50 in rabbits, mice (mg/kg):
0.25 i.v.; 7-10 s.c. (Saunders, Stacey).Derivative
Ethyl ester.
Nomenclature
CAS number: 459-72-3
C
4H
7FO
2; mol wt 106.10.
C 45.28%, H 6.65%, F 17.91%, O 30.16%.
Properties
Liq, odor of ethyl acetate. d20.5 1.0926. bp758 121.6°. nD20.5 1.3767. Sol in water.Caution
Fluoroacetates are highly toxic.
Potential symptoms of overexposure are vomiting; apprehension; clonic-tonic
convulsions; depression; loss of consciousness; metabolic acidosis;
hypotension; cardiac arrhythmias; respiratory failure; hypocalcemia;
renal failure; death (Goncharov).Use
Rodenticide.
