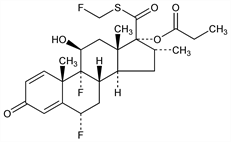
4211. Fluticasone Propionate
Nomenclature
Description and references
Derivative of flumethasone, q.v. Prepn: NL 81 00707; G. H. Phillipps et al., US 4335121 (1981, 1982 both to Glaxo). Series of articles on structure-activity, pharmacology and clinical studies: Respir. Med. 84, Suppl. A, 19-35 (1990). Clinical trials in allergic rhinitis: E. O. Meltzer et al., J. Allergy Clin. Immunol. 86, 221 (1990). Evaluation in untreated celiac disease: H. C. Mitchison et al., Gut 32, 260 (1991). Clinical trials in COPD: P. L. Paggiaro et al., Lancet 351, 773 (1998); P. S. Burge et al., Br. Med. J. 320, 1297 (2000). Clinical comparison with zafirlukast, in persistant asthma: J. H. Brabson et al., Am. J. Med. 113, 15 (2002). Clinical trial in asthma in adults: M. Masoli et al., Eur. Respir. J. 28, 960 (2006); in children: R. L. Wasserman et al., Ann. Allergy Asthma Immunol. 96, 808 (2006).
Properties
Crystals, mp 272-273° (dec). [α]D +30° (c = 0.35). Practically insol in water. Freely sol in DMSO, DMF; slightly sol in methanol, 95% ethanol.Therapeutic Category
Antiallergic; antiasthmatic; anti-inflammatory.
Therapeutic Category (Veterinary)
Antihistaminic; anti-inflammatory.Keywords
Antiallergic (Steroidal, Nasal); Glucocorticoid; Antiasthmatic (Steroidal, Inhalant)