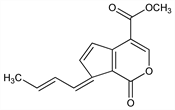
4285. Fulvoplumierin
Nomenclature
CAS number: 20867-01-0
(7E)-7-(2E)-Butenylidene-1,7-dihydro-1-oxocyclopenta[c]pyran-4-carboxylic acid methyl ester; 3-(2-butenylidene)-2-carboxy-α-(hydroxymethylene)-1,4-cyclopentadiene-1-acetic
acid δ-lactone methyl ester; methyl 7-crotonylidenecyclopenta[c]pyran-1-(7H)-one-4-carboxylate. C14H12O4; mol wt 244.24.
C 68.85%, H 4.95%, O 26.20%.
Description and references
Occurs together with plumieride and plumericin. Isoln from Plumeria acutifolia Poir., Apocynaceae, also from roots of P. rubra var alba: Grumbach et al., Experientia 8, 224 (1952). Structure: Schmid, Bencze, Helv. Chim. Acta 36, 206, 1468 (1953). Stereochemistry: Albers-Schnberg et al., ibid. 45, 1406 (1962). Synthesis: Büchi, Carlson, J. Am. Chem. Soc. 90, 5336 (1968); 91, 6470 (1969).