4317. Fusidic Acid
Nomenclature
Description and references
Antibiotic isolated from the fermentation broth of Fusidium coccineum; structurally similar to cephalosporin P1, q.v. Inhibits bacterial protein synthesis by interference with elongation factor G. Isoln and structure: W. O. Godtfredsen et al., Nature 193, 987 (1962); Lancet I, 928 (1962); W. O. Godtfredsen, S. Vangedal, Tetrahedron 18, 1029 (1962). Identity with ramycin: H. Vanderhaeghe et al., Nature 205, 710 (1965). Structure: D. Arigoni et al., Experientia 19, 521 (1963). Stereochemistry: W. O. Godtfredsen et al., Tetrahedron 21, 3505 (1965). Synthetic studies: W. G. Dauben et al., J. Am. Chem. Soc. 94, 8593 (1972); R. E. Ireland, U. Hengartner, ibid. 3652; M. Tanabe et al., Tetrahedron Lett. 1977, 1481. Total synthesis: W. G. Dauben et al., J. Am. Chem. Soc. 104, 303 (1982). Review of structure-activity relationships: W. von Daehne et al., Adv. Appl. Microbiol. 25, 95-146 (1979). Series of articles on pharmacology and clinical experience: Int. J. Antimicrob. Agents 12, Suppl. 2, S1-S93 (1999). Review of use in Staphylococcus aureus infections: D. Dobie, J. Gray, Arch. Dis. Child. 89, 74-77 (2004).
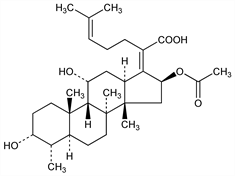
Properties
Crystals from ether or benzene, mp 192-193°. [α]D20 9° (chloroform). uv max: 204 nm (ε 9900). pK: 5.35 in water. Sol in alc, acetone, chloroform, pyridine, dioxane; sparingly sol in water, ether, hexane. LD50 in mice (g/kg): 1.2 s.c.; 1.5 orally (Godtfredsen).Derivative
Sodium salt.Nomenclature
Properties
Crystals, sol in water. LD50 in mice (g/kg): 0.2 i.v. (Godtfredsen, Nature 1962).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics)