4318. Fustin
Nomenclature
CAS number: 20725-03-5
(2R,3R)-rel-2-(3,4-Dihydroxyphenyl)-2,3-dihydro-3,7-dihydroxy-4H-1-benzopyran-4-one; 3,3′,4′,7-tetrahydroxyflavanone; dihydrofisetin. C15H12O6; mol wt 288.25.
C 62.50%, H 4.20%, O 33.30%.
Description and references
From wood of Rhus cotinus L. (Venice sumac) and R. succedanea L., Anacardiaceae: Schmid, Ber. 19, 1734 (1886); from Gleditsia triacanthos L., Leguminosae: Chadenson et al., Compt. Rend. 249, 1362 (1955). Structure: Oyamada, Ann. 538, 44 (1939). Stereochemistry: Weinges, Ann. 627, 229 (1959); Roux, Paulus, Biochem. J. 77, 315 (1960); Gaffield, Tetrahedron 26, 4093 (1970).
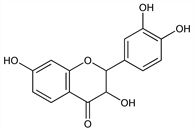
Derivative
(±)-Form.Properties
Crystals, mp 226-228°. [α]D21 2.4°.Derivative
(±)-Form tetraacetate.C23H20O10; mol wt 456.40.
C 60.53%, H 4.42%, O 35.06%.