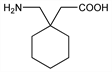
4319. Gabapentin
Nomenclature
Description and references
Calcium channel ligand structurally related to γ-aminobutyric acid (GABA), q.v., designed to cross the blood brain barrier. Prepn: G. Satzinger et al., DE 2460891 (1976 to Gdecke); eidem, US 4024175 (1977 to Warner-Lambert). Pharmacokinetics and metabolism: K.-O. Vollmer et al., Arzneim.-Forsch. 36, 830 (1986). Clinical pharmacology: B. Saletu et al., Int. J. Clin. Pharmacol. Ther. Toxicol. 24, 362 (1986). GC determn in biological fluids: W. D. Hooper et al., J. Chromatogr. 529, 167 (1990). LC/MS/MS determn in plasma: N. V. S. Ramakrishna et al., J. Pharm. Biomed. Anal. 40, 360 (2006). Clinical trial in treatment of pain in diabetic neuropathy: M. Backonja et al., J. Am. Med. Assoc. 280, 1831 (1998); in treatment of postherpetic neuralgia: M. Rowbotham et al., ibid. 1837. Review of pharmacology and clinical trials in epilepsy: B. Schmidt in Antiepileptic Drugs, R. H. Levy et al., Eds. (Raven Press, New York, 3rd ed., 1989) pp 925-935; K. L. Goa, E. M. Sorkin, Drugs 46, 409-427 (1993); of pharmacology and use in pain management: M. A. Rose, P. C. A. Kam, Anaesthesia 57, 451-462 (2002); of interactions with voltage-gated calcium channels: K. G. Sutton, T. P. Snutch, Drug Dev. Res. 54, 167-172 (2002); of clinical experience in postoperative pain: K.-Y. Ho et al., Pain 126, 91-101 (2006); of mechanism of action: J.-K. Cheng, L.-C. Chiou, J. Pharm. Sci. 100, 471-486 (2006).
Properties
Crystals from ethanol/ether, mp 162-166° (Satzinger); also reported as mp 165-167° (Schmidt). pKa1 (25°) 3.68; pKa2 10.70. Isoelectric point 7.14. Partition coefficient (octanol/buffer): 0.075 (pH 7.4). Solubility in water at pH 7.4 exceeds 10%.Therapeutic Category
Anticonvulsant; analgesic.
Keywords
Anticonvulsant; Analgesic; Non-opioids