4321. Gaboxadol
Nomenclature
Description and references
Selective extrasynaptic GABA agonist (SEGA); structural analog of muscimol, q.v. Prepn: P. Krogsgaard-Larsen, Acta Chem. Scand. B 31, 584 (1977); idem, EP 167; idem, US 4278676 (1979, 1981 both to H. Lundbeck & Co.). GABA agonist effects: idem et al., Nature 268, 53 (1977). 3H-THIP binding study: E. Falch, P. Krogsgaard-Larsen, J. Neurochem. 38, 1123 (1982). HPLC determn: S. M. Madsen, J. Chromatogr. 238, 509 (1982). Pharmacokinetics: idem et al., Acta Pharmacol. Toxicol. 53, 353 (1983). Pharmacodynamics and potential therapeutic uses: A. V. Christensen et al., Pharm. Weekbl. Sci. Ed. 4, 145 (1982). Clinical effects on nocturnal sleep and hormone secretion: M. Lancel et al., Am. J. Physiol. Endocrinol. Metab. 281, E130 (2001); on night sleep and cognitive performance: S. Mathias et al., Neuropsychopharmacology 30, 833 (2005). Review of clinical development: R. Huckle, Curr. Opin. Investig. Drugs 5, 766-773 (2004).
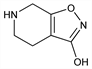
Properties
Colorless cryst, mp 242-244° (dec). uv max (methanol): 212 nm (log ε 3.64). pKa (water, 25°): 4.44 ±0.03; 8.48 ±0.04.Derivative
Hydrobromide.Nomenclature
Properties
Faintly reddish cryst from methanol-ether, mp 162-163° (dec). LD50 in mice (mg/kg): 80 i.v., 145 i.p.; >320 orally (U.S. patent).Derivative
Hydrochloride.Nomenclature
Use
As a molecular probe to study GABA receptors.Therapeutic Category
Sedative, hypnotic.
Keywords
Sedative/Hypnotic