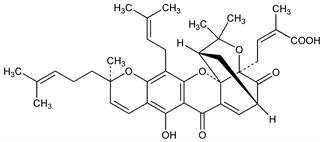
4361. Gambogic Acid
Nomenclature
Description and references
Cytotoxic principle of gamboge, q.v., a gum-resin from the latex of garcinia trees, especially Garcinia hanburyi Hook. f., Guttiferae. Isoln: M. Amorosa, L. Lipparini, Ann. Chim. (Rome) 45, 977 (1955). Structure: W. D. Ollis et al., Tetrahedron 21, 1453 (1965); S. A. Ahmad et al., J. Chem. Soc. C 1966, 772; G. Cardillo, L. Merlini, Tetrahedron Lett. 8, 2529 (1967). HPLC determn in plasma: L. Ding et al., J. Chromatogr. B 846, 112 (2007). Apoptosis inducing activity: H.-Z. Zhang et al., Bioorg. Med. Chem. 12, 309 (2004). Antiangiogenic activity: T. Yi et al., Cancer Res. 68, 1843 (2008). Toxicology: Q. Guo et al., Basic Clin. Pharmacol. Toxicol. 99, 178 (2006).
Properties
Yellow-orange solid. [α]D20 685° (methanol). uv max (ethanol): 217, 280, 291, 362 nm (ε 26000, 16700, 17000, 14900). Sol in DMSO, ethanol. LD50 i.p. in mice: 45.9 mg/kg (Guo).Derivative
Methyl ester monomethyl ether.Nomenclature
Properties
Yellow prisms from methanol, mp 130-131°. [α]D22 560° (c = 0.7 in chloroform). uv max (ethanol): 224, 299 nm (ε 36000, 13600).Derivative
Pyridine salt.Nomenclature
Properties
Orange needles from ether + petr ether, mp 147-149°. [α]D 550° (chloroform). uv max (ethanol): 291.5, 359.5 nm (ε 22300, 18100).Derivative
Amide.Nomenclature
Description and references
Prepn: S. X. Cai et al., WO 00044216; eidem, US 6462041 (2000, 2002 both to Cytovia). Neurotrophic activity: S.-W. Jang et al., Proc. Natl. Acad. Sci. USA 104, 16329 (2007).