4363. Ganciclovir
Nomenclature
Description and references
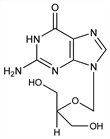
Nucleoside analog structurally related to acyclovir, q.v. Prepn: J. P. Verheyden, J. C. Martin, US 4355032 (1982 to Syntex); K. K. Ogilvie et al., Can. J. Chem. 60, 3005 (1982); W. T. Ashton et al., Biochem. Biophys. Res. Commun. 108, 1716 (1982); J. C. Martin et al., J. Med. Chem. 26, 759 (1983). Antiviral spectrum in vitro: K. O. Smith et al., Antimicrob. Agents Chemother. 22, 55 (1982). Mode of action study: Y.-C. Cheng et al., Proc. Natl. Acad. Sci. USA 80, 2767 (1983). Clinical treatment of cytomegalovirus infection in immunodeficient patients: S. H. Koretz et al., N. Engl. J. Med. 314, 801 (1986). Symposium on pharmacology and clinical efficacy vs cytomegalovirus: Rev. Infect. Dis. 10, Suppl. 3, S457-S572 (1988). Review: C. S. Crumpacker, N. Engl. J. Med. 335, 721-729 (1996).
Properties
Crystals from methanol, mp 250° (dec) (Verheyden, Martin); also reported as crystalline monohydrate from water, mp 248-249° (dec) (Ashton); crystals from water, mp >300° (Martin). uv max (methanol): 254 nm (ε 12880). Soly in water (25°): 4.3 mg/ml at pH 7. LD50 i.p. in mice: 1-2 g/kg (Martin).Derivative
Sodium salt.Nomenclature
Therapeutic Category
Antiviral.
Keywords
Antiviral; Nucleosides/Nucleotides