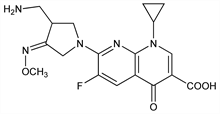
4389. Gemifloxacin
Nomenclature
Description and references
Third generation fluorinated quinolone antibacterial. Prepn: J. H. Kwak et al., EP 688772; C. Y. Hong et al., US 5633262 (1995, 1997 both to LG Chemical); idem et al., J. Med. Chem. 40, 3584 (1997); of methanesulfonate: A. R. Kim et al., WO 9842705 (1998 to LG Chemical). Antibacterial spectrum in vitro: R. Wise, J. M. Andrews, J. Antimicrob. Chemother. 44, 679 (1999). LC/MS/MS determn in human plasma: E. Doyle et al., J. Chromatogr. B 746, 191 (2000). Clinical pharmacokinetics: A. Allen et al., Antimicrob. Agents Chemother. 44, 1604 (2000). Review of antibacterial activity, pharmacology, and clinical trials: J. M. Blondeau, B. Missaghi, Expert Opin. Pharmacother. 5, 1117-1152 (2004).
Properties
Off-white, amorphous solid from chloroform-ethanol as the monohydrate, mp 235-237°.Derivative
Methanesulfonate.Nomenclature
Properties
White to light brown solid, mp 195° (dec). Soly in water at 25° (mg/ml): 162 (pH 2); 19 (pH 6); 0.5 (pH 7).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs