4403. Geraniol
Nomenclature
CAS number: 106-24-1
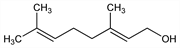
(E)-3,7-Dimethyl-2,6-octadien-1-ol; trans-3,7-dimethyl-2,6-octadien-8-ol; lemonol. C10H18O; mol wt 154.25.
C 77.87%, H 11.76%, O 10.37%.
Description and references
An olefinic terpene alcohol constituting the chief part of oil of rose and oil of palmarosa; also found in many other essential oils such as citronella, lemon grass, etc. Isomeric with linalool. Isoln: Jacobsen, Ann. 157, 234 (1871). Structure: Verley, Bull. Soc. Chim. Fr. 25, 68 (1919); J. L. Simonsen, The Terpenes vol. I (University Press, Cambridge, 2nd ed., 1947) pp 40-52. Stereochemistry: Burrell et al., Proc. Chem. Soc. London 1959, 263; Bates et al., J. Org. Chem. 28, 1086 (1963). Synthesis: Burrell et al., J. Chem. Soc. C 1966, 2144; K. Takabe et al., Chem. Lett. 1977, 1025; K. K. Mathew et al., Indian J. Chem. B20, 340 (1981).
Properties
Oily liq. Sweet rose odor. bp757 229-230°; bp12 114-115°. d420 0.8894. nD20 1.4766. uv max: 190-195 nm (ε 18000). Practically insol in water. Miscible with alcohol, ether.Derivative
Acetate.C12H20O2; mol wt 196.29.
C 73.43%, H 10.27%, O 16.30%.
Properties
Sweet, fragrant liq. bp ≈242° with decompn. d1515 0.9174. nD15 1.4628. Almost insol in water. Very sol in alcohol; miscible with ether.Derivative
Butyrate.C14H24O2; mol wt 224.34.
C 74.95%, H 10.78%, O 14.26%.
Properties
Liquid. Characteristic fragrant odor. bp18 152°. d417 0.901. Almost insol in water. Sol in alc, ether.Derivative
Formate.C11H18O2; mol wt 182.26.
C 72.49%, H 9.95%, O 17.56%.