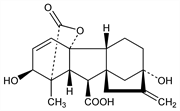
4419. Gibberellic Acid
Nomenclature
Description and references
Plant hormone; most outstanding of the plant-growth promoting metabolites of Gibberella fujikuroi. Isoln: P. J. Curtis, B. E. Cross, Chem. Ind. (London) 1954, 1066; B. E. Cross, J. Chem. Soc. 1954, 4670; P. W. Brian et al., US 2842051; C. T. Calam, P. J. Curtis, US 2950288; A. J. Birch et al., US 2977285 (1958, 1960, 1961, all to ICI). Stereochemistry and structure: G. Stork, H. Newman, J. Am. Chem. Soc. 81, 5518 (1959); B. E. Cross et al., Proc. Chem. Soc. London 1959, 302; F. McCapra et al., ibid. 1962, 185; D. C. Aldridge et al., J. Chem. Soc. 1963, 143; P. M. Bourn et al., ibid. 1963, 154. Partial synthesis: E. J. Corey et al., J. Am. Chem. Soc. 93, 7316 (1971). Stereospecific total synthesis: eidem, ibid. 100, 8034 (1978). Promotes growth of seedlings: M. J. Bukovac, S. H. Wittwer, Q. Bull. Mich. Agric. Exp. Stn. 39, 307 (1956); J. M. Merritt, J. Agric. Food Chem. 6, 184 (1958). Reviews: B. E. Cross et al. in Adv. Chem. Ser. 28, entitled “Gibberellins,” J. M. Merritt, Ed. (ACS, Washington DC, 1961) p 13; series of articles in Plant Growth Subst., Proc. 7th Int. Conf., D. J. Carr, Ed. (Springer-Verlag, Berlin, 1972).