4429. Girard Reagents
Description and references
Quaternary ammonium acetylhydrazine chlorides which form water-soluble hydrazones with carbonyl compounds. The hydrazones formed can subsequently be hydrolyzed in order to regenerate the original carbonyl compounds. Prepn: A. Girard, G. Sandulesco, GB 6640 (1934); eidem, Helv. Chim. Acta 19, 1095 (1936); A. Girard, Org. Synth. coll. vol. II, 85 (1943). Review: Wheeler, Chem. Rev. 62, 205 (1962). Use of Girard reagents in separation of carbonyl compounds: Schubert, Wehrberger, Endokrinologie 48, 70 (1965); R. E. J. Mitchel, H. C. Birnboim, Anal. Biochem. 81, 47 (1977); W. Holstein, D. Severin, Erdoel Kohle, Erdgas, Petrochem. 32, 487 (1979); C.A. 92, 44207b (1980).
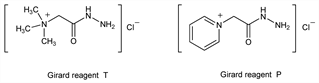
Derivative
Girard reagent T.Nomenclature
CAS number: 123-46-6
2-Hydrazinyl-N,N,N-trimethyl-2-oxoethanaminium chloride
(1:1); (carboxymethyl)trimethylammonium chloride hydrazide; betaine hydrazide hydrochloride. C5H14ClN3O; mol wt 167.64.
C 35.82%, H 8.42%, Cl 21.15%, N 25.07%, O 9.54%.
Properties
Highly hygroscopic needles. May be stored in well-stoppered containers. Material that has developed an odor should be recrystallized from abs ethanol. mp 192° (slight decomp). Very freely sol in water; sol in about 150 parts of absolute ethanol; more sol in methanol. Also very sol in acetic acid, glycerol, ethylene glycol. Practically insol in organic solvents devoid of hydroxyl groups.Derivative
Girard reagent P.Nomenclature
CAS number: 1126-58-5
1-(2-Hydrazinyl-2-oxoethyl)pyridinium chloride (1:1); 1-(carboxymethyl)pyridinium chloride hydrazide. C7H10ClN3O; mol wt 187.63.
C 44.81%, H 5.37%, Cl 18.90%, N 22.40%, O 8.53%.