4440. Glimepiride
Nomenclature
Description and references
Oral sulfonylurea; promotes insulin release at ATP-sensitive potassium channels on pancreatic β-cells via binding to a 65 kDa subunit of the sulfonylurea receptor. Prepn: R. Weyer et al., DE 2951135; eidem, US 4379785 (1981, 1983 both to Hoechst). Synthesis: R. Weyer, V. Hitzel, Arzneim.-Forsch. 38, 1079 (1988). Pharmacology: K. Geisen, ibid. 1120. Effects on insulin and glucagon secretion: V. Leclercq-Meyer et al., Biochem. Pharmacol. 42, 1634 (1991). HPLC determn in plasma: Y.-K. Song et al., J. Chromatogr. B 810, 143 (2004). Clinical pharmacokinetics: K. Ratheiser et al., Arzneim.-Forsch. 43, 856 (1993). Toxicity study: U. Schollmeier et al., ibid. 1038. Series of articles on pharmacology and clinical efficacy: Diabetes Res. Clin. Pract. 28 Suppl., S115-S149 (1995). Review of pharmacology and clinical use in type 2 diabetes: H. D. Langtry, J. A. Balfour, Drugs 55, 563-584 (1998); A. L. McCall, Expert Opin. Pharmacother. 2, 699-713 (2001).
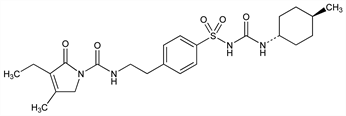
Properties
White to yellowish-white crystalline powder. mp 207°. Practically insol in water. Sol in DMSO; slightly sol in acetone; very slightly sol in acetonitrile, methanol.Therapeutic Category
Antidiabetic.
Therapeutic Category (Veterinary)
Antidiabetic.Keywords
Antidiabetic; Sulfonylurea Derivatives