4442. Glipizide
Nomenclature
Description and references
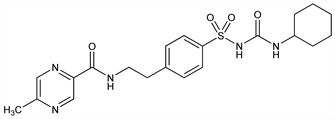
Second generation sulfonylurea with hypoglycemic activity. Prepn: V. Ambrogi, W. Logemann, DE 2012138; eidem, US 3669966 (1970, 1972 both to Carlo Erba); Ambrogi et al., Arzneim.-Forsch. 21, 200 (1971). Pharmacology: eidem, ibid. 208; Marigo et al., ibid. 215. Metabolism: Goldaniga et al., ibid. 23, 242 (1973); Fuccella et al., J. Clin. Pharmacol. 13, 68 (1973). Pharmacokinetics and pharmacodynamics of extended-release formulation: M. Chung et al., J. Clin. Pharmacol. 42, 651 (2002). Evaluation in diabetic cats: E. C. Feldman et al., J. Am. Vet. Med. Assoc. 210, 772 (1997). Clinical evaluation in diabetes: D. C. Simonson et al., Diabetes Care 20, 597 (1997). Toxicity: Ambrogi et al., Arzneim.-Forsch. 21, 208 (1971). Review of pharmacology and therapeutic efficacy: R. N. Brogden et al., Drugs 18, 329-353 (1979); H. E. Lebovitz, Pharmacotherapy 5, 63-77 (1985).
Properties
Crystals from ethanol, mp 208-209°. Also reported as mp 200-203°. pKa 5.9 Insol in water, alcohols. Sol in 0.1 N NaOH; freely sol in DMF. LD50 in mice, rats (g/kg): >3, 1.2 i.p. (Ambrogi).Therapeutic Category
Antidiabetic.
Therapeutic Category (Veterinary)
Antidiabetic.Keywords
Antidiabetic; Sulfonylurea Derivatives