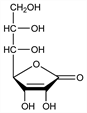
4451. d-Glucoascorbic Acid
Nomenclature
CAS number: 528-88-1
d-arabino-Hept-2-enonic acid γ-lactone; 3-keto-d-glucoheptonofuranolactone. C7H10O7; mol wt 206.15.
C 40.78%, H 4.89%, O 54.33%.
Description and references
A physiologically inactive homolog of ascorbic acid. Prepn: Ault et al., J. Chem. Soc. 1933, 741; Baird et al., ibid. 1934, 62; Reichstein et al., Helv. Chim. Acta 17, 510 (1934); Stacey, Turton, J. Chem. Soc. 1946, 661; Stedehouder, Rec. Trav. Chim. 71, 831 (1952). Review on analogs of ascorbic acid: Smith, Adv. Carbohydr. Chem. 2, 79 (1946).