4484. Glycerol
Nomenclature
Description and references
Obtained from oils and fats as byproduct in the manuf of soaps and fatty acids. During World War I supplementary quantities were produced by the “Protol” fermentation process from sugar, a process based upon the fixation of acetaldehyde by sodium sulfite. Just prior to World War II the synthesis of glycerol from propylene was announced. Production from sugars by fermentation: Onishi, US 3012945 (1961 to Noda). Identity with incorporation factor: Kuehl et al., J. Am. Chem. Soc. 82, 2079 (1960). In nucleic acid the incorporation factor may exist as a bound form of glycerol. Acute toxicity: W. Bartsch et al., Arzneim.-Forsch. 26, 1581 (1976). Reviews and bibliographies: J. W. Lawrie, Glycerol and the Glycols (New York, 1928); G. Leffingwell, M. Lesser, Glycerin (Brooklyn, 1945); C. S. Miner, N. N. Dalton, Glycerol (New York, 1953); C. Lüttgen, Glyzerin und glyzerinhnliche Stoffe (Heidelberg, 2nd ed., 1955); J. C. Kern in Kirk-Othmer Encyclopedia of Chemical Technology vol. 11 (Wiley-Interscience, New York, 3rd ed., 1980) pp 921-932.
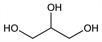
Properties
Syrupy liquid. Sweet warm taste. About 0.6 times as sweet as cane sugar. Hygroscopic; also absorbs H2S, HCN, SO2. Contact with strong oxidizing agents such as chromium trioxide, potassium chlorate, or potassium permanganate may produce an explosion. Neutral to litmus. Solidifies after prolonged cooling at 0° forming shiny orthorhombic crystals, mp 17.8°. bp760 290.0° (dec); bp400 263.0°; bp200 240.0°; bp100 220.1°; bp60 208.0°; bp20 182.2°; bp10 167.2°; bp5 153.8°; bp1.0 125.5°. nD15 1.4758; nD20 1.4746; nD25 1.4730. d1515 1.26557; d2020 1.26362; d2525 1.26201. Flash point, open cup: 350°F (176°C). Specific gravities of 95% aq soln w/w (U.S.P. grade): d1515 1.25270; d2020 1.25075; d2525 1.24910; 90% aq soln w/w: d1515 1.23950; d2020 1.23755; d2525 1.23585; 80% d1515 1.213; 70% d1515 1.185; 60% d1515 1.157; 50% d1515 1.129; 20% d1515 1.049; 5% d1515 1.0122. Viscosity (cP at 20°): 5% soln 1.143; 10% 1.311; 25% 2.095; 50% 6.050; 60% 10.96; 70% 22.94; 83% 111. Freezing points of aq solns w/w: 10% 1.6°; 30% 9.5°; 50% 23.0°; 66.7% 46.5°; 80% 20.3°; 90% 1.6°. Miscible with water, alcohol. One part dissolves in 11 parts ethyl acetate, in about 500 parts ethyl ether. Insol in benzene, chloroform, carbon tetrachloride, carbon disulfide, petr ether, oils. LD50 in rats (ml/kg): >20 orally; 4.4 i.v. (Bartsch).Caution
Potential symptoms of overexposure to mist are irritation of eyes, skin, respiratory system; headache, nausea, vomiting; kidney injury. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 152.Use
As solvent, humectant, plasticizer, emollient, sweetener, in the manuf of nitroglycerol (dynamite), cosmetics, liq soaps, liqueurs, confectioneries, blacking, printing and copying inks, lubricants, elastic glues, lead oxide cements; to keep fabrics pliable; to preserve printing on cotton; for printing rollers, hectographs; to keep frost from windshields; as antifreeze in automobiles, gas meters and hydraulic jacks, in shock absorber fluids. In fermentation nutrients in the production of antibiotics. Titration reagent. In tests for heavy metals. Pharmaceutic aid (humectant; solvent, vehicle). Leffingwell and Lesser (op. cit.) give 1583 different uses.Therapeutic Category
Diagnostic aid (ophthalmic).
Therapeutic Category (Veterinary)
Emollient, demulcent. As a source of glucose in bovine ketosis.Keywords
Diagnostic Aid