4483. Glyceric Acid
Nomenclature
CAS number: 473-81-4
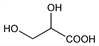
2,3-Dihydroxypropanoic acid; α,β-dihydroxypropionic acid; dl-glyceric acid. C3H6O4; mol wt 106.08.
C 33.97%, H 5.70%, O 60.33%.
Description and references
Prepn from α,β-dibromopropionic acid by treatment with silver oxide: Karrer, Klarer, Helv. Chim. Acta 7, 931 (1924); from isoserine and nitrous acid: Fischer, Jacobs, Ber. 40, 1069 (1907); from glycerol and nitrous acid: Mulder, Ber. 9, 1902 (1876); Beilstein, Ann. 120, 229 (1861). Dextrorotatory glyceric acid is obtained by the action of Penicillia or Aspergilli on the dl-form: McKenzie, Harden, J. Chem. Soc. 83, 431 (1903). Levorotatory glyceric acid has been obtained by the oxidation of d(+)-glyceric aldehyde: Wohl, Schellenberg, Ber. 55, 1408 (1922).
Properties
Syrup, dec on distn. pK (25°): 3.55. Miscible with water, alcohol, acetone. Nearly insol in ether.Derivative
l(+)-Form.Nomenclature
CAS number: 28305-26-2
(2S)-2,3-Dihydroxypropanoic acid. Properties
Syrup. Its esters and salts are levorotatory and its salts are much more sol in water than those of the dl-form.Derivative
l-Form calcium salt.Nomenclature
CAS number: 6057-35-8
Ca(C3H5O4)2.2H2O; mol wt 286.25.
Ca 14.00%, C 25.18%, H 4.93%, O 55.89%.
Properties
Monoclinic sphenoidal crystals, mp 137°. One gram dissolves in 10 ml water. [α]D20 14.6° (c = 5).Derivative
d()-Form.Nomenclature
CAS number: 6000-40-4
(2R)-2,3-Dihydroxypropanoic acid. Properties
Syrup. Its salts are dextrorotatory.Derivative
d-Form calcium salt.Nomenclature
CAS number: 6000-41-5
Ca(C3H5O4)2.2H2O; mol wt 286.25.
Ca 14.00%, C 25.18%, H 4.93%, O 55.89%.