4486. Glycerophosphoric Acid
Nomenclature
CAS number: 57-03-4 (dl-α-form);
17181-54-3
(β-form)
Phosphoric acid glycerol esters. C3H9O6P; mol wt 172.07.
C 20.94%, H 5.27%, O 55.79%, P 18.00%.
Description and references
Three isomers exist: β-glycerophosphoric acid, the d(+)- and l()-forms of α-glycerophosphoric acid. The l-α-acid is the naturally occurring form; the β-acid, present in hydrolyzates of lecithins from natural sources, arises from migration of the phosphoryl group from the α-carbon atom. See review: Dawson, Annu. Rep. Prog. Chem. 55, 365 (1958). Prepn by phosphorylation of glycerol results in a mixture of the α- and β-acids: Cherbuliez, Weniger, Helv. Chim. Acta 29, 2006 (1946). Prepn and configuration of l-α-acid: Baer, Fischer, J. Biol. Chem. 128, 491 (1939). Prepn of d-α-acid: eidem, ibid. 135, 321 (1940). Separation of α-acid from β- and polyglycerophosphoric acids: Carrara, IT 460219 (1950), C.A. 46, 5077a (1952).
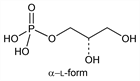
Properties
Absolute acid (commercial mixture of α- and β-acids); clear syrupy liq, mp 25°. d414 1.59. Tends to dec during concn; hence, usually marketed as a 25-50% soln. Soluble in water, alcohol.Derivative
α-Acid l-form.Nomenclature
CAS number: 5746-57-6
(2S)-1-(Dihydrogen phosphate)-1,2,3-propanetriol.