4528. Gossypol
Nomenclature
Description and references
Yellow pigment found in cottonseed; functions as a natural insecticide in plants. Name derived from the botanical name of the cotton plant, Gossypium L., Malvaceae. Isoln: J. Longmore, J. Soc. Chem. Ind. 5, 200 (1886); L. Marchlewski, J. Prakt. Chem. 60, 84 (1899); K. N. Campbell et al., J. Am. Chem. Soc. 59, 1723 (1937). Structural studies: R. Adams et al., ibid. 60, 2193 (1938). Synthesis: Edwards, J. Am. Oil Chem. Soc. 47, 441 (1970). NMR studies: J. W. Jaroszewski et al., NMR Spectrosc. Drug Res. 26, 75 (1988). Vibrational CD structural studies: T. B. Freedman et al., Chirality 15, 196 (2003). Prepn of enantiomers: M. K. Dowd, ibid. 486. HPLC determn: G. B. Marcelle et al., J. Pharm. Sci. 73, 396 (1984). Metabolism studies: M. B. Abou-Donia et al., Lipids 5, 938 (1970). Mechanism of action study: C.-Y. G. Lee et al., Mol. Cell. Biochem. 47, 65 (1982). Structure-activity study in tumor cell lines: M. D. Shelley et al., Anti-Cancer Drugs 11, 209 (2000). Clinical evaluation in malignant glioma: P. Bushunow et al., J. Neuro-Oncol. 43, 79 (1999); in male contraception: E. M. Coutinho et al., Contraception 61, 61 (2000). Review of chemistry: R. Adams et al., Chem. Rev. 60, 555-574 (1960); of toxicity in livestock: S. E. Morgan, Vet. Clin. North Am. Food Anim. Pract. 5, 251-262 (1989); of clinical pharmacology and use as a male contraceptive agent: D. Wu, Drugs 38, 333-341 (1989); E. M. Coutinho, Contraception 65, 259-263 (2002).
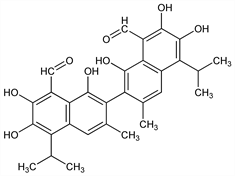
Properties
Exists in 3 tautomeric forms. Yellow crystals from ether, mp 184°; from chloroform, mp 199°; from ligroin, mp 214°. Light sensitive. Very sol in methanol, ethanol, cold dioxane, diethylene glycol, ether, ethyl acetate, acetone, carbon tetrachloride, pyridine, chloroform, DMF, lipids. Freely sol (with slow decompn) in dil aq solns of ammonia and sodium carbonate. Slightly sol in glycerol, cyclohexane. Insol in water. Absorption max: 385 nm (ε 18000).Derivative
R-Form.Nomenclature
Properties
Bright yellow crystals from acetone, mp 181-184° (dec). [α]D28 386 to 390° (c = 0.5 g/100 g soln in CHCl3) (Dowd).Derivative
S-Form.Nomenclature
Description and references
Isoln from Thespesia populnea: T. J. King, L. B. de Silva, Tetrahedron Lett. 9, 261 (1968); S. C. Datta et al., Indian J. Chem. 10, 263 (1972). Synthesis: A. I. Meyers, J. J. Willemsen, Tetrahedron 54, 10493 (1998).