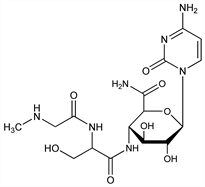
4529. Gougerotin
Nomenclature
Description and references
Antibiotic substance with antibacterial and antineoplastic activity. Isoln from Streptomyces gougerotii: Kanzaki et al., J. Antibiot. 15A, 93 (1962). Identity with asteromycin: Ikeuchi et al., ibid. 25, 548 (1972). Structure: Iwasaki, Yakugaku Zasshi 82, 1358 (1962). Revised structure: Fox et al., Tetrahedron Lett. 1968, 6029; Watanabe et al., Chem. Pharm. Bull. 17, 416 (1969). Total synthesis: eidem, J. Am. Chem. Soc. 94, 3272 (1972); Lichtenthaler et al., Tetrahedron Lett. 1975, 3527. Identity with aspiculamycin: Lichtenthaler et al., ibid. 665. Mechanism of action study: J. C. Lacal et al., J. Antibiot. 33, 441 (1980). Reviews: Clark in Antibiotics vol. 1, D. Gottlieb, P. D. Shaw, Eds. (Springer-Verlag, New York, 1967) pp 278-282; Yukioka, ibid. vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (1975) pp 448-458.