4533. Gramine
Nomenclature
CAS number: 87-52-5
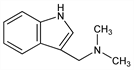
N,N-Dimethyl-1H-indole-3-methanamine; 3-(dimethylaminomethyl)indole; Donaxine. C11H14N2; mol wt 174.24.
C 75.83%, H 8.10%, N 16.08%.
Description and references
In chlorophyll-deficient mutants of barley: Euler et al., Z. Physiol. Chem. 217, 23 (1933). In the Asiatic reed Arundo donax L., Gramineae: Orechoff, Norkina, Ber. 68, 436 (1935). From Acer saccharinum L. (the Silver Maple) and A. rubrum L., Aceraceae: Pachter et al., J. Org. Chem. 24, 1285 (1959); Pachter, J. Am. Pharm. Assoc. Sci. Ed. 48, 670 (1959). Synthesis: Kühn, Stein, Ber. 70, 567 (1937). Biosynthesis from tryptophan in barley: Bowden, Marion, Can. J. Chem. 29, 1037 (1951); O'Donovan, Leete, J. Am. Chem. Soc. 85, 461 (1963); Gower, Leete, ibid. 3683; see also Gross et al., Tetrahedron Lett. 1971, 4047.
Properties
Shiny, flat needles or plates from acetone, mp 138-139°. Absorption spectrum: Kanakoa et al., Chem. Pharm. Bull. 8, 294 (1960). Sol in alcohol, ether, chloroform; slightly sol in cold acetone. Practically insol in petr ether, water.Derivative
Hydrochloride.C11H14N2.HCl; mol wt 210.70.
C 62.70%, H 7.18%, N 13.30%, Cl 16.83%.
Properties
Crystals from ethanol + ether, dec 191°. Sol in water.Derivative
Methiodide.C11H14N2.CH3I; mol wt 316.18.
C 45.58%, H 5.42%, N 8.86%, I 40.14%.