4558. Guanabenz
Nomenclature
Description and references
α2-Adrenergic agonist. Prepn: J. Yates, E. Haddock, GB 1019120 (1966 to Shell), C.A. 64, 11132h (1966). Use as antihypertensive: W. J. Houlihan et al., DE 1804634 (1969 to Sandoz), C.A. 71, 89976j (1969). Pharmacology: T. Baum et al., J. Pharmacol. Exp. Ther. 171, 276 (1970); E. Lampa et al., Experientia 36, 228 (1980). Disposition of 14C-guanabenz in humans: R. H. Meacham et al., Clin. Pharmacol. Ther. 27, 44 (1980). Mechanism of action: G. F. DiBona, J. Cardiovasc. Pharmacol. 6, Suppl. 3, S543 (1984). Radioimmunoassay determn in plasma: H. Tatsumi, Arzneim.-Forsch. 34, 1704 (1984). Clinical studies: A. Reppelli et al., Boll. Soc. Ital. Cardiol. 23, 177 (1978); C. V. Ram et al., J. Clin. Pharmacol. 19, 148 (1979). Clinical studies in opiate withdrawal: F. S. Tennant, R. A. Rawson, NIDA Res. Monogr. Ser. 49, 338 (1984); J. T. Murphy, Drug Intell. Clin. Pharm. 19, 32 (1985). Review of pharmacodynamic properties and therapeutic efficacy: B. Holmes et al., Drugs 26, 212-229 (1983). Comprehensive description: C. M. Shearer, Anal. Profiles Drug Subs. 15, 319-336 (1986).
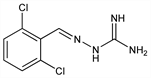
Properties
White solid from acetonitrile, mp 227-229° (dec).Derivative
Monoacetate.Nomenclature
Properties
Solid, mp 192.5° (dec). Soly at 25° (mg/ml): water 11; alcohol 50; propylene glycol 100; chloroform 0.6; ethyl acetate 1.Therapeutic Category
Antihypertensive.
Keywords
α-Adrenergic Agonist; Antihypertensive; Guanidine Derivatives