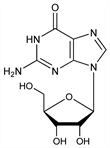
4566. Guanosine
Nomenclature
Description and references
Constituent of nucleic acids. Prepn from yeast nucleic acid: P. A. Levene, L. W. Bass, Nucleic Acids (New York, 1931) p 163. Prepn from polynucleotides: P. A. Levene, E. Jorpes, J. Biol. Chem. 86, 389 (1930). Prepn from plants: H. Stendel, E. Peiser in G. Klein, Handbuch der Pflanzenanalyse IV (Vienna, 1933) p 448. Structure: Levene, Tipson, J. Biol. Chem. 97, 491 (1932); Gulland et al., J. Chem. Soc. 1934, 1639; Tsuboi et al., Biochim. Biophys. Acta 55, 1 (1962). Synthesis: Davoll, J. Chem. Soc. 1958, 1593. Tautomerism in aq soln: Miles et al., Science 142, 1458 (1963). Crystal structure and conformation: Bugg et al., Biochem. Biophys. Res. Commun. 3, 436 (1968). Review: Basic Principles in Nucleic Acid Chemistry vol. 1, P. O. P. Ts'o, Ed. (Academic Press, New York, 1974) passim.