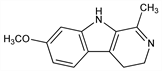
4613. Harmaline
Nomenclature
CAS number: 304-21-2
4,9-Dihydro-7-methoxy-1-methyl-3H-pyrido[3,4-b]indole; 1-methyl-7-methoxy-3,4-dihydro-β-carboline; 3,4-dihydroharmine; harmidine; harmalol methyl ether; O-methylharmalol. C13H14N2O; mol wt 214.26.
C 72.87%, H 6.59%, N 13.07%, O 7.47%.
Description and references
CNS stimulant from seeds of Peganum harmala L., Zygophyllaceae: Goebel, Ann. 38, 363 (1841); from Banisteria caapi Spruce, Malpighiaceae: Hochstein, Paradies, J. Am. Chem. Soc. 79, 5735 (1957). Structure: Manske et al., J. Chem. Soc. 1927, 1. Synthesis: Spth, Lederer, Ber. 63, 120, 2102 (1930); Spenser, Can. J. Chem. 37, 1851 (1959). Identity with harmidine: Robinson, Chem. Ind. (London) 1965, 605. Pharmacology: Fuentes, Longo, Neuropharmacology 10, 15 (1971). Metabolism: Ho et al., Biochem. Pharmacol. 20, 1313 (1971). Review of structure and synthesis work: Hofmann, Sven. Farm. Tidskr. 75, 933 (1971), C.A. 76, 149609g (1972).