4614. Harmalol
Nomenclature
CAS number: 525-57-5
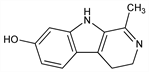
4,9-Dihydro-1-methyl-3H-pyrido[3,4-b]indol-7-ol; 3,4-dihydro-1-methyl-9H-pyrido[3,4-b]indol-7-ol. C12H12N2O; mol wt 200.24.
C 71.98%, H 6.04%, N 13.99%, O 7.99%.
Description and references
From seeds of Peganum harmala L., Zygophyllaceae: Gbel, Ann. 38, 363 (1841); Fischer, Ber. 18, 400 (1885); by demethylation of harmaline: Coulthard et al., Biochem. J. 27, 727 (1933). HPLC determn in Peganum harmala L. seeds: M. Kartal et al., J. Pharm. Biomed. Anal. 31, 263 (2003). In vitro neuroprotective effects: D. H. Kim et al., Eur. J. Neurosci. 13, 1861 (2001).
Derivative
Trihydrate.Nomenclature
CAS number: 6027-99-2
Properties
Red needles from water. Dec 212° (anhydr). Readily sol in hot water, acetone, chloroform, alkali hydroxides, but not carbonates. Aq solns are yellow with green fluorescence.Derivative
Lactate.Nomenclature
CAS number: 6028-08-6
C12H12N2O.C3H6O3.
Properties
mp 174-176°; monohydrate as bright yellow leaflets, mp 116-120°. Sol in water and alcohol.Derivative
O-Ethylharmalol.Nomenclature
CAS number: 6010-17-9
C14H16N2O; mol wt 228.29.
C 73.66%, H 7.06%, N 12.27%, O 7.01%.
Properties
Brown needles from alc, mp 237-239°.Derivative
O-n-Propylharmalol.Nomenclature
CAS number: 6028-01-9
C15H18N2O; mol wt 242.32.
C 74.35%, H 7.49%, N 11.56%, O 6.60%.
Properties
Brown needles from alc, mp 195-197°.Derivative
O-n-Butylharmalol.Nomenclature
CAS number: 6197-42-8
C16H20N2O; mol wt 256.34.
C 74.97%, H 7.86%, N 10.93%, O 6.24%.