4615. Harman
Nomenclature
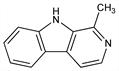
CAS number: 486-84-0
1-Methyl-9H-pyrido[3,4-b]indole; 3-methyl-4-carboline; 2-methyl-β-carboline; aribine; loturine; passiflorin. C12H10N2; mol wt 182.22.
C 79.10%, H 5.53%, N 15.37%.
Description and references
From bark of Sickingia rubra (Mart.) K. Schum. (Arariba rubra Mart.), Rubiaceae; Symplocus racemosa Roxb., Symplocaceae; and Passiflora incarnata L., Passifloraceae: Rieth, Wohler, Ann. 120, 247 (1861); Spth, Monatsh. Chem. 40, 351; idem, ibid. 41, 401 (1920); Neu, Arzneim.-Forsch. 4, 601 (1954). Structure: Neu, ibid. 6, 94 (1956). Synthesis: Harvey, Robson, J. Chem. Soc. 1938, 97; Snyder et al., J. Am. Chem. Soc. 70, 222 (1948); Clemo, Holt, J. Chem. Soc. 1953, 1313; Kametani et al., J. Chem. Soc. C 1968, 1006. Isoln from cigarette smoke: Poindexter, Carpenter, Chem. Ind. (London) 1962, 176. Toxicity study: E. B. Sigg et al., Arch. Int. Pharmacodyn. 149, 164 (1964).
Properties
Bitter orthorhombic crystals from heptane + cyclohexane, mp 237-238°. Exhibits bright blue fluorescence in uv light. pKa: 7.37, 14.6. uv max (methanol): 234, 287, 347 nm (log ε 4.57, 4.21, 3.66). Absorption and fluorescence spectra: O. S. Wolfbeis et al., Monatsh. Chem. 113, 509 (1982). Practically insol in water. Sol in dil acids. LD50 i.p. in mice: 50 mg/kg (Sigg).Derivative
Hydrochloride.C12H10N2.HCl; mol wt 218.68.
C 65.91%, H 5.07%, N 12.81%, Cl 16.21%.