4672. Hetacillin
Nomenclature
CAS number: 3511-16-8
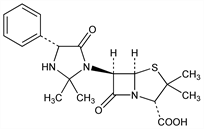
(2S,5R,6R)-6-[(4R)-2,2-Dimethyl-5-oxo-4-phenyl-1-imidazolidinyl]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 6-(2,2-dimethyl-5-oxo-4-phenyl-1-imidazolidinyl)penicillanic
acid; phenazacillin; BRL-804; Versapen (BMS). C19H23N3O4S; mol wt 389.47.
C 58.59%, H 5.95%, N 10.79%, O 16.43%, S 8.23%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn and structure: Hardcastle et al., J. Org. Chem. 31, 897 (1966); Johnson, Panetta, US 3198804 (1965 to Bristol-Myers). Pharmacology: Kirby, Kind, Ann. N.Y. Acad. Sci. 145, 291 (1967); Ueda et al., J. Antibiot. 20B, 206 (1967), C.A. 69, 95016w (1968). Stability studies: Saccani, Pansera, Boll. Chim. Farm. 107, 640 (1968). Epimerization at C-6 to epihetacillin: Johnson et al., Tetrahedron Lett. 1968, 1903.
Properties
Rectangular plates from water + methyl isobutyl ketone, dec 182.8-183.9°; also reported as mp 189.2-191.0°. [α]D25 +366° (pyridine). Practically insol in most organic solvents and water; sol in dil aq NaOH soln (pH 7-8), DMF, DMSO, pyridine, methanol (with dec).Derivative
Potassium salt.Nomenclature
CAS number: 5321-32-4
Hetacin-K (Fort Dodge); Natacillin (Banyu); Versatrex (BMS). C19H22KN3O4S; mol wt 427.56.
C 53.37%, H 5.19%, K 9.14%, N 9.83%, O 14.97%, S 7.50%.
Derivative
Methyl ester.C20H25N3O4S; mol wt 403.50.
C 59.53%, H 6.24%, N 10.41%, O 15.86%, S 7.95%.
Properties
Crystals from carbon tetrachloride, mp 101.5-102°. Sol in most organic solvents.Derivative
Epihetacillin.Properties
Crystals, mp 164-165°. [α]D23 +232° (pyridine).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins