4701. Hexestrol
Nomenclature
CAS number: 84-16-2
4,4′-(1,2-Diethyl-1,2-ethanediyl)bisphenol; 4,4′-(1,2-diethylethylene)diphenol; meso-3,4-bis(p-hydroxyphenyl)-n-hexane; 4,4′-dihydroxy-γ,δ-diphenylhexane; 4,4′-dihydroxy-α,β-diethyldiphenylethane; dihydrodiethylstilbestrol; hexoestrol; Synthovo; Cycloestrol; Hexanoestrol; Hormoestrol; Syntrogène. C18H22O2; mol wt 270.37.
C 79.96%, H 8.20%, O 11.84%.
Description and references
From anethole HBr: Campbell et al., Proc. Roy. Soc. B128, 253 (1940); Bernstein, Wallis, J. Am. Chem. Soc. 62, 2871 (1940); US 2357985 (1944); Buu-Hoi, Hoán, J. Org. Chem. 14, 1023 (1949). By the reduction of Grignard compds with cobaltous chloride: Kharasch et al., J. Am. Chem. Soc. 65, 491 (1943). Proof of meso configuration: Wessely, Welleba, Ber. 74, 777 (1941). Comprehensive description: H. Y. Aboul-Enein et al., Anal. Profiles Drug Subs. 11, 347-374 (1982).
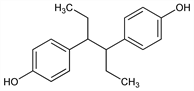
Properties
Needles from benzene, thin plates from dil alc, mp 185-188°. Freely sol in ether; sol in acetone, alcohol, methanol; slightly sol in benzene, chloroform. Sol in vegetable oils upon slight warming, also in dil solns of alkali hydroxides. Practically insol in water and in dil mineral acids.Derivative
Diacetate.Nomenclature
Retalon-Lingual.Properties
Crystals, mp 137-139°.Derivative
Dipropionate.Nomenclature
Retalon Oleosum.Properties
Crystals from petr ether, mp 127-128°.Derivative
Diphosphate.Nomenclature
CAS number: 14188-82-0
Hexestrol 4,4′-diphosphoric ester; Cytostatin. C18H24O8P2; mol wt 430.33.
C 50.24%, H 5.62%, O 29.74%, P 14.40%.
Description and references
Prepn: GB 593480 (1947 to Roche); Atherton et al., US 2490573 (1949 to Hoffmann-La Roche).
Therapeutic Category
Estrogen; antineoplastic (hormonal).
Therapeutic Category (Veterinary)
Estrogen.Keywords
Antineoplastic (Hormonal); Estrogens; Estrogen; Nonsteroidal