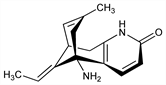
4755. Huperzine A
Nomenclature
Description and references
Reversible alkaloid inhibitor of AChE which crosses the blood-brain barrier. Occurs as the ()-form in the vegetative part of clubmosses; teas brewed from these mosses have traditionally been used in China to alleviate memory problems. Isolated as selagine from Lycopodium selago L., Lycopodiaceae: J. Muszynski, Q. J. Pharm. Pharmacol. 21, 34 (1948); from Huperzia serrata as huperzine: Chin. Co-op Res. Group, J. Tradit. Chin. Med. 2, 45 (1982). Original structure: Z. Valenta et al., Tetrahedron Lett. 1960, 26; revised structure as huperzine A: J.-S. Liu et al., Can. J. Chem. 64, 837 1986. Identity with selagine: W. A. Ayer et al., ibid. 67, 1538 (1989). Synthesis of (±)-form: A. P. Kozikowski et al., J. Org. Chem. 56, 4636 (1991); G. Campiani et al., ibid. 58, 7660 (1993). NMR spectra: B. N. Zhou et al., Phytochemistry 34, 1425 (1993). HPLC determn in serum: J. Ye et al., J. Chromatogr. B 817, 187 (2005). Anticholinesterase activity: Y.-E. Wang et al., Acta Pharmacol. Sin. 7, 110 (1986); binding profile of enantiomers: M. McKinney et al., Eur. J. Pharmacol. 203, 303 (1991); binding specificity: Y. Ashani et al., Mol. Pharmacol. 45, 555 (1994). Clinical evaluation in senile dementia: R.-W. Zhang et al., Acta Pharmacol. Sin. 12, 259 (1991). Brief review of chemistry and clinical use: D. Bai, Pure Appl. Chem. 65, 1103-1112 (1993).
Properties
Monoclinic crystals from acetone, mp 214-215°. [α]D 147° (c = 0.36 in CH3OH) (Ayer). Also reported as mp 230°. [α]D24.5 150.4° (c = 0.498 in MeOH) (Liu). uv max (EtOH): 231, 313 nm (log ε 4.01, 3.89).Therapeutic Category
In treatment of memory disorders.