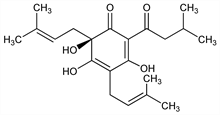
4754. Humulon
Nomenclature
CAS number: 26472-41-3
(6R)-3,5,6-Trihydroxy-4,6-bis(3-methyl-2-butenyl)-2-(3-methyl-1-oxobutyl)-2,4-cyclohexadien-1-one; α-bitter acid; α-lupulic acid; humulone. C21H30O5; mol wt 362.46.
C 69.59%, H 8.34%, O 22.07%.
Description and references
Antibiotic constituent of hops (Humulus lupulus L., Moraceae). See also Lupulon. Isoln from commercial hops: Bungener, Bull. Soc. Chim. [2] 45, 487 (1886); Barth, Lintner, Ber. 31, 2022 (1898); Wollmer, Ber. 49, 780 (1916); Lewis et al., J. Clin. Invest. 28, 916 (1949). Structure: Riedl, Ber. 85, 692 (1952); Carson, J. Am. Chem. Soc. 73, 4652 (1951). Absolute configuration and structure of preferred isomer: DeKeukeleire, Verzele, Tetrahedron 26, 385 (1970).