4756. Hyalobiuronic Acid
Nomenclature
CAS number: 499-15-0
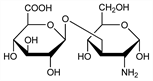
2-Amino-2-deoxy-3-O-β-d-glucopyranuronosyl-d-glucose; 3-O-(β-d-glucopyranosyluronic acid)-2-amino-2-deoxy-d-glucose. C12H21NO11; mol wt 355.30.
C 40.57%, H 5.96%, N 3.94%, O 49.53%.
Description and references
Disaccharide unit of hyaluronic acid. Isoln from hyaluronic acid: Rapport et al., Nature 168, 996 (1951). Structure: Weissman, Meyer, J. Am. Chem. Soc. 76, 1753 (1954). Synthesis: Takanashi et al., ibid. 84, 3029 (1962).
Properties
Rectangular prisms from water, darken at 190° with no characteristic melting or dec point. pK1′ = 2.6, pK2′ = 7.1. Shows mutarotation: [α]D20 +34° → +30° (c = 1.08 in 0.1N HCl). Sparingly sol in hot water, dilute HCl, dil NaHCO3. Practically insol in water, glacial acetic acid, ethanol, methanol and pyridine.Derivative
N-Acetylhyalobiuronic acid.C14H23NO12; mol wt 397.33.
C 42.32%, H 5.83%, N 3.53%, O 48.32%.